Clinical Drug Development: Clinical Trial Methodologies and Innovations
- HMX

Learn how therapeutics are assessed during clinical drug development.
- Online; Instructor-Paced
$1,025
Multi-course bundle pricing is also available across enrollment periods.
Certificate
Earn a Certificate of Completion upon successfully completing all coursework.
10 Weeks, 1-2 Hours/Week
Most people can expect to spend around 15–20 hours total, but this depends on your baseline knowledge and how carefully you take notes.
- Online; Instructor-Paced
$1,025
Multi-course bundle pricing is also available across enrollment periods.
Certificate
Earn a Certificate of Completion upon successfully completing all coursework.
10 Weeks, 1-2 Hours/Week
Most people can expect to spend around 15–20 hours total, but this depends on your baseline knowledge and how carefully you take notes.
On This Page
Overview
Clinical drug development has evolved dramatically over time and continues to advance today. With new techniques being introduced, the design and statistical aspects have become more sophisticated in parallel with the increased use of technology and the introduction of new drug modalities. The rigor and discipline of this process means that people can trust that the medicines they take are likely to be safe and effective and that beneficial treatments can make their way through the process in a safe and timely manner.
Gain insights into the methodologies of clinical trials and the future of clinical drug development with this advanced HMX course. Through an engaging, online curriculum, you will learn how therapeutics are assessed during clinical drug development. Explore aspects of conducting clinical trials, including epidemiological considerations, trial design strategies, statistical methods for data analysis, and oversight and review processes.
Learning Objectives
- Learn some of the key epidemiological considerations that need to be taken into account when designing and conducting a clinical trial
- Understand how a clinical trial is designed, carried out, interpreted, and presented
- Learn about the process of drug review and the regulatory steps that are taken to move a drug from clinical drug development to the drug market for use in patients
Overview
Clinical drug development has evolved dramatically over time and continues to advance today. With new techniques being introduced, the design and statistical aspects have become more sophisticated in parallel with the increased use of technology and the introduction of new drug modalities. The rigor and discipline of this process means that people can trust that the medicines they take are likely to be safe and effective and that beneficial treatments can make their way through the process in a safe and timely manner.
Gain insights into the methodologies of clinical trials and the future of clinical drug development with this advanced HMX course. Through an engaging, online curriculum, you will learn how therapeutics are assessed during clinical drug development. Explore aspects of conducting clinical trials, including epidemiological considerations, trial design strategies, statistical methods for data analysis, and oversight and review processes.
Learning Objectives
- Learn some of the key epidemiological considerations that need to be taken into account when designing and conducting a clinical trial
- Understand how a clinical trial is designed, carried out, interpreted, and presented
- Learn about the process of drug review and the regulatory steps that are taken to move a drug from clinical drug development to the drug market for use in patients
Questions?
Have questions about HMX courses, enrollment, or learning options? Contact our team by email—we’re happy to assist.
About the Course
HMX online courses bring complex material to life through engaging biomedical visualizations, clinical applications, and true-to-life scenarios paired with lectures from Harvard Medical School faculty and leading experts.
Who Should Enroll
This advanced course is appropriate for pharmaceutical professionals or researchers seeking a firm understanding of the process of clinical drug development following preclinical research; professionals working in a related sector and would like to update knowledge of the latest advancements in clinical trial methodologies; health care professionals looking to advance your expertise in clinical drug development and trial design.
Course Format
Most people can expect to spend around 15–20 hours total, but this depends on your baseline knowledge, how carefully you take notes, and how seriously you take the assessments. Lessons from the courses are released toward the beginning of the course session and remain available until the courses end, so you can work at your own pace. There is a final exam at the end of each course that can be taken at any time during the multi-week final exam period. The flexible course format makes this an ideal choice for working professionals.
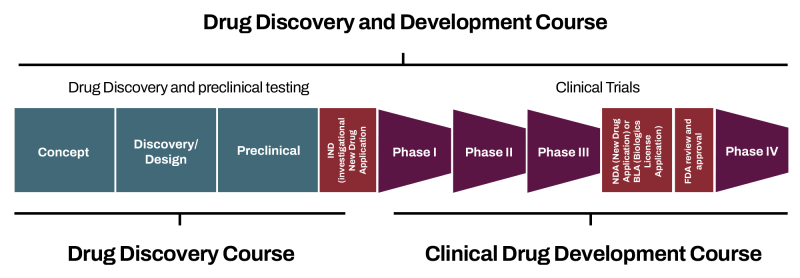
Which Drug Discovery/Development Course Should I Take?
The progression of drug development is divided into distinct phases. This course, Clinical Drug Development: Clinical Trial Methodologies and Innovations, focuses on clinical trials delving into phases I-IV in greater detail. The overall end-to-end process is explained at a high level in the Drug Discovery and Development: Overview of the End-to-End Therapeutic Development Pipeline course. The Drug Discovery: Approaches to Discovering and Designing Different Therapeutic Modalities course dives into the upfront stages in more detail including discovery, design and preclinical research.
Group Enrollments
HMX courses are ideal for organizations looking to train teams or larger groups. Group pricing is available, making it a cost-effective investment in team development.
Faculty
- Lecturer in Biological Chemistry and Molecular Pharmacology | Harvard Medical School

Paul Ridker
MD, MPH
- Eugene Braunwald Professor of Medicine, Harvard Medical School
- Senior Physician, Brigham and Women's Hospital / Director of the Center for Cardiovascular Disease Prevention, Brigham and Women's Hospital
Scott Berry, PhD, President and Senior Statistical Scientist, Berry Consultants
Julie Buring, ScD, Professor of Medicine, Harvard Medical School / Epidemiologist, Brigham and Women’s Hospital
Rory Collins, FRS, FMedSci, Professor of Medicine and Epidemiology, University of Oxford / Principal Investigator and Chief Executive, UK Biobank
George Q. Daley, MD, PhD, Dean of the Faculty of Medicine, Harvard University / Dean of Harvard Medical School / Caroline Shields Walker Professor of Medicine, Robert A. Stranahan Professor of Pediatrics, and Professor of Biological Chemistry and Molecular Pharmacology, Harvard Medical School / Investigator, Howard Hughes Medical Institute / Director of the Stem Cell Transplantation Program, Boston Children’s Hospital
Margaret Hamburg, MD, Foreign Secretary, National Academy of Medicine / President, American Association for the Advancement of Science / Former Commissioner, FDA / Harvard Medical School alumna
Joe Miletich, MD, PhD, Senior Vice President of Discovery, Preclinical, and Early Development, Merck
Marc Pfeffer, MD, PhD, Victor J. Dzau Distinguished Professor of Medicine, Harvard Medical School / Cardiovascular Physician, Brigham and Women’s Hospital
Alice Shaw, MD, PhD, Professor of Medicine, Harvard Medical School / Thoracic Oncologist, Massachusetts General Hospital
Priya Singhal, MD, MPH, Senior Vice President and Head of Global Safety & Regulatory Sciences, Biogen / Harvard Medical School alumna
Meredith Buxton, PhD, MPH, President and CEO of Global Coalition for Adaptive Research
Brian Alexander, MD, MPH, CEO of Foundation Medicine / Associate Professor of Radiation Oncology, Harvard Medical School / Disease Center Leader of the Center for Neuro-Oncology, Dana–Farber Cancer / Institute Director of Therapeutic Science, Harvard Medical School
Michelle Albert, MD, MPH, Associate Dean of Admissions, Professor of Medicine, and Walter A. Haas-Lucie Stern Endowed Chair in Cardiology, University of California, San Francisco / President of the American Heart Association
ADDITIONAL CONTRIBUTORS
Juli Coraor Fried, MD/PhD candidate, Harvard Medical School
Hannah Hoffman, MD/PhD candidate, Harvard Medical School
Course Quiz
Are you ready for an advanced course? We recommend knowledge of basic chemistry, biology, and physics, as well as knowledge of key pharmacology concepts. Gauge your knowledge of pharmacology by taking this short quiz.
From Our Learners
Hear from HMX students as they share their experiences learning online with Harvard Medical School.
"I found the real-world examples of clinical trials, presented by top investigators who had been part of them, to be the most exciting course content."
Request Information
Interested in learning more about this program? Sign up for details.



